Research
In Khaperskyy lab, we study virus-host interactions and cellular stress responses.
Our goal is to understand how cells respond to virus infection and other stresses to identify molecular pathways that can be targeted for host-directed antiviral therapies and cancer treatments.
Below are research themes in the lab
Stress Granules
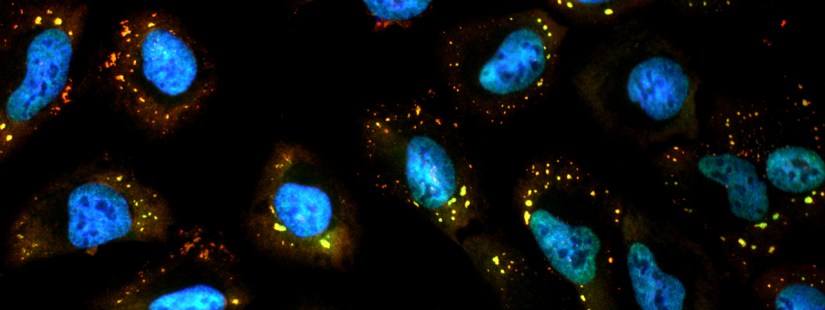
Stress granules (SGs) are cytoplasmic foci of RNA and proteins that form in cells under stress. Several research projects in the laboratory study properties and functions of these granules. SGs are best detected via microscopy using immunofluorescence (IF) staining for SG proteins or fluorescence in situ hybridization (FISH) of RNAs. In our laboratory we do… Continue reading Stress Granules
Host shutoff

To gain preferential access to the protein synthesis machinery and to disrupt induction of antiviral responses by infected cell many viruses block host gene expression. This blockade is called host shutoff and it is mediated by viral factors that either destroy host messenger RNAs (mRNAs) or interfere with their synthesis. Influenza A virus (IAV) encodes… Continue reading Host shutoff
Antiviral responses
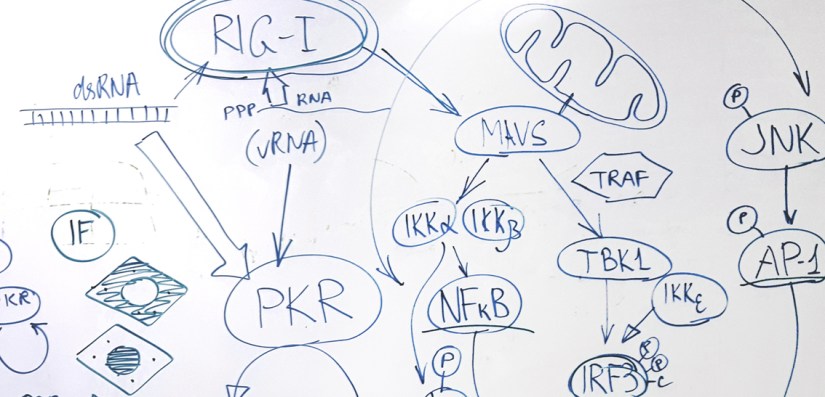
Intrinsic immune responses are the cell’s first line of defense against invading pathogens. In virus-infected cells these responses are activated following detection of pathogen-associated molecular patterns (PAMPs) that are normally absent from uninfected cells (e.g. viral genomic RNA). One of the activated responses is the transient translation arrest. This arrest interferes with viral gene expression… Continue reading Antiviral responses

